 
Biotechnology companies are also using clonoSEQ to measure the effectiveness of their therapies in clinical trials. Physicians use it to monitor disease progression, remission, and relapse in response to therapy. IM Revenue Forecast (Author's estimates based on ADPT filings)ĪDPT is also leveraging its technology in the molecular diagnostics space and currently markets clonoSEQ, a Minimal Residual Disease "MRD" used to monitor cancer prevalence in the body. Given the uncertainty surrounding the outcome of ADPT's collaboration with Roche, our DCF model incorporates an initial decline in Collaboration Revenue of 15% in 2023, followed by a gradual rebound at an annual rate of 10% through 2027. In 2023, management expects IM revenue to be $92 million - $96 million, representing -6% to -1.2% change in the segment's sales due to an expected decline in amortized revenue from Roche's 2019 pre-payment, partially offset by growth in revenue from Biopharma Services. In 2022, ADPT reported an 11% increase in revenue related to its drug-development business (Roche and Biopharma Services Revenue.) Biopharma Services increased 30%, compared to a modest contribution from Roche, whose initial development phase is ending. Although likely less than the Roche partnership, ADPT is entitled to milestone payments related to these drug development projects. This business model decreases operating costs and risk, providing an immediate source of income. Currently, immunoSEQ is used in 140 clinical studies conducted by 85 biotech companies. 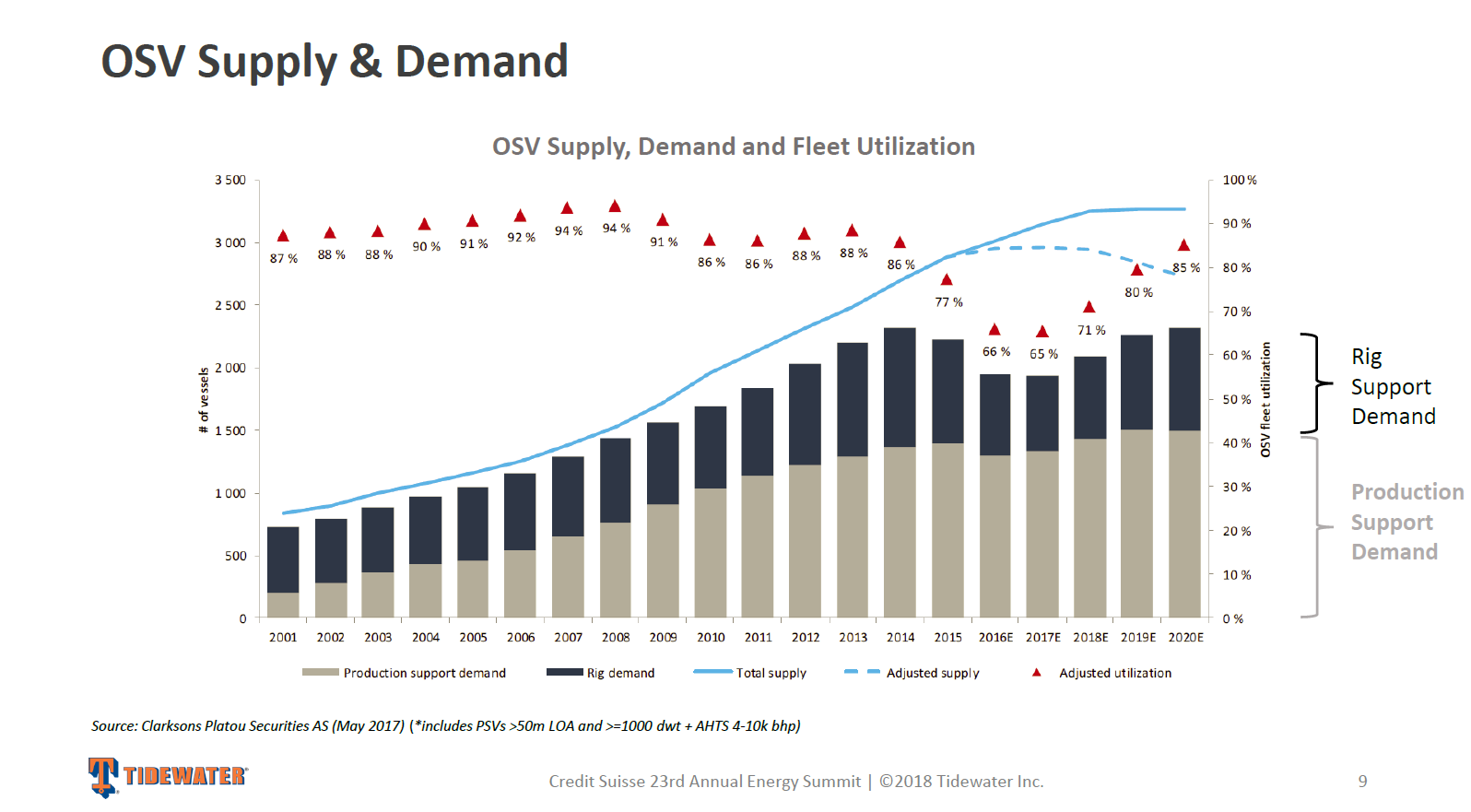
Instead of monopolizing its technology, ADPT commercialized its Intellectual Property as an immuno-sequencing service under the immunoSEQ brand to pharmaceutical companies exploring the potential of developing therapies using the body's immune system. In addition, we are separately able to receive tiered royalties at a rate ranging from the mid-single digits to the mid-teens on aggregate worldwide net sales of products arising from the strategic collaboration, subject to certain reductions, with aggregate minimum floors. Subsequent to receipt of regulatory approval in January 2019, we received a non-refundable, upfront payment of $300.0 million in February 2019 and may be eligible to receive more than $1.8 billion over time, including payments of up to $75.0 million upon the achievement of specified regulatory milestones, up to $300.0 million upon the achievement of specified development milestones and up to $1,430.0 million upon the achievement of specified commercial milestones. In December 2018, we entered into a worldwide collaboration and license agreement with Genentech (the "Genentech Agreement") to leverage our capability to develop cellular therapies in oncology. Chad Robins, Q4 2022 Earnings CallĪDPT is entitled to royalty and milestone payments related to the Roche partnerships, constituting a lucrative but highly uncertain revenue potential in the future, conditional to the success of the drug development venture. We're making good progress with Genentech on two cell therapy programs. 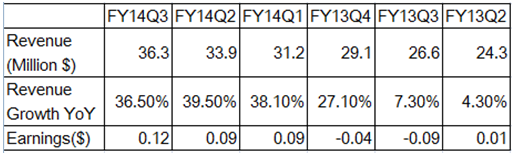
Revenue DriversĪDPT is developing two drugs with Genentech, a subsidy of Roche's ( OTCQX:RHHBY), acting as the technology provider for the two projects in return for a $300 million upfront payment received in 2019, to be recognized in installments as the project progresses. Our hold rating mirrors our desire to see consistent operating leverage such as the one demonstrated in Q4. In the following paragraphs, we explore ADPT's current product lines and future market prospects. Management at ADPT has calculated the total addressable markets in each segment, as shown below. Although the latter has a far lesser financial prospect than drug development, it is the closest that ADPT can be to the clinical applications of its technology at the present time. In spite of this, the company has been able to generate revenue from its immunoSEQ platform by forming strategic alliances and seizing opportunities in the relatively untapped molecular diagnostics sector. The technology is still in its very early stages, and there are several hurdles to overcome before it can be implemented in the clinic. By contrast, an allergic reaction, which berserkly activates the immune system, could kill a person within minutes.Īdaptive Biotechnologies ( NASDAQ: ADPT), along with its partners, is trying to explore the potential for harnessing the natural immune system to treat diseases. 
An untreated Ebola patient will likely die within six to sixteen days. The immune system is so powerful that our bodies spend more energy keeping it in check than fighting disease. Chawalit Banpot/iStock via Getty Images Investment Thesis 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
